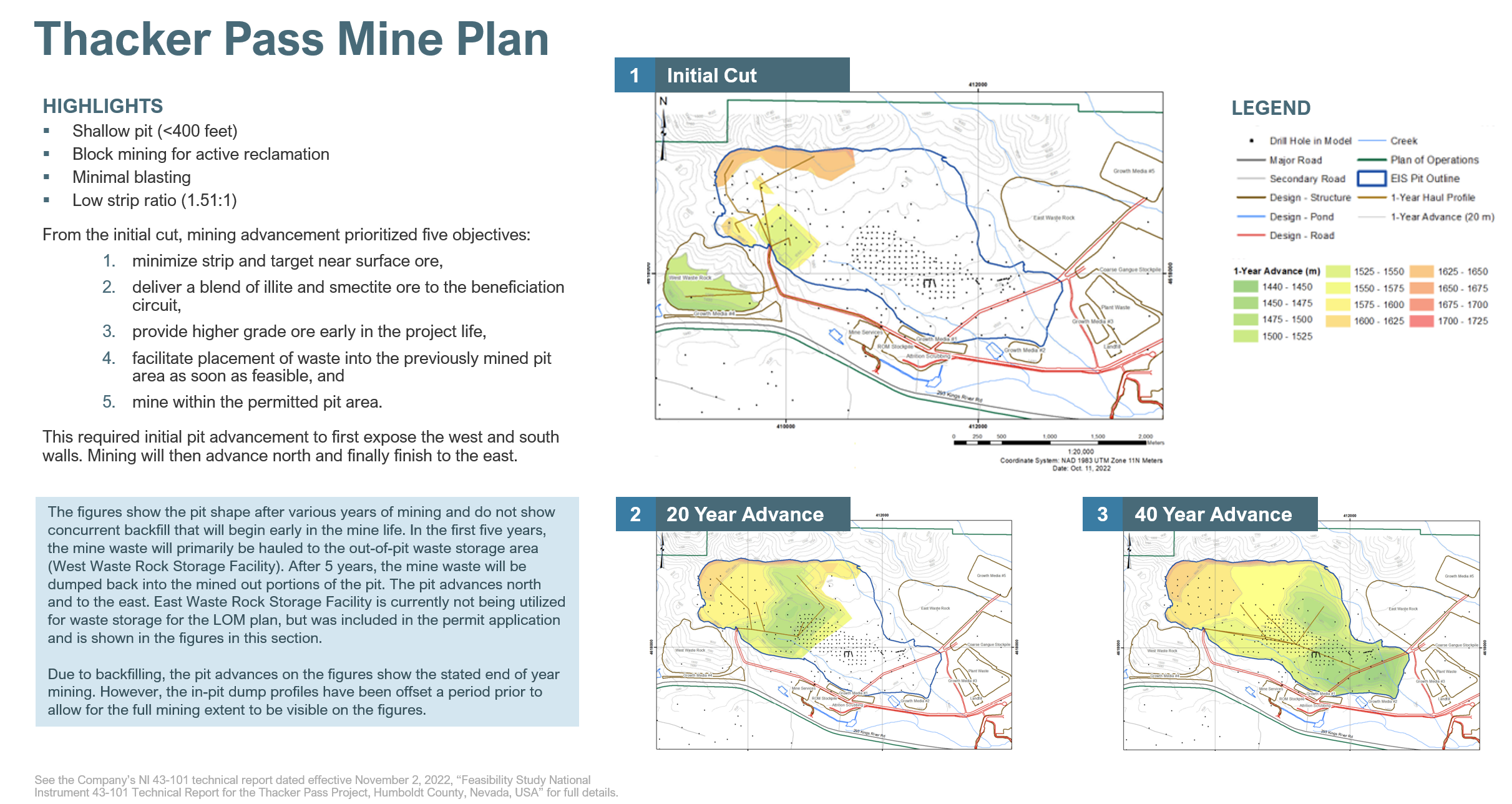
The sedimentary-clay deposit at Thacker Pass will use similar mining techniques to many coal deposits. The shallow open-pit will have an average depth of 300 feet and will be mined in blocks in order to be actively reclaimed. Once one block is mined to the bottom, mining operations will move to the next block, while backfilling begins in the previously mined area. The backfilled pits will then be capped with topsoil and revegetated with native species.
Block mining allows us to reclaim the land quickly and only have a fraction of the pit exposed at any given time. This will minimize long-term visual impacts and allow for substantial restoration of pre-project land uses by wildlife.
 (Opens in new window)
(Opens in new window)
The production process is designed to use conventional and commonly-available equipment, arranged to take advantage of the distinctive qualities of the high-grade ore. The process comprises a series of steps to concentrate, separate and produce battery-quality lithium chemicals.
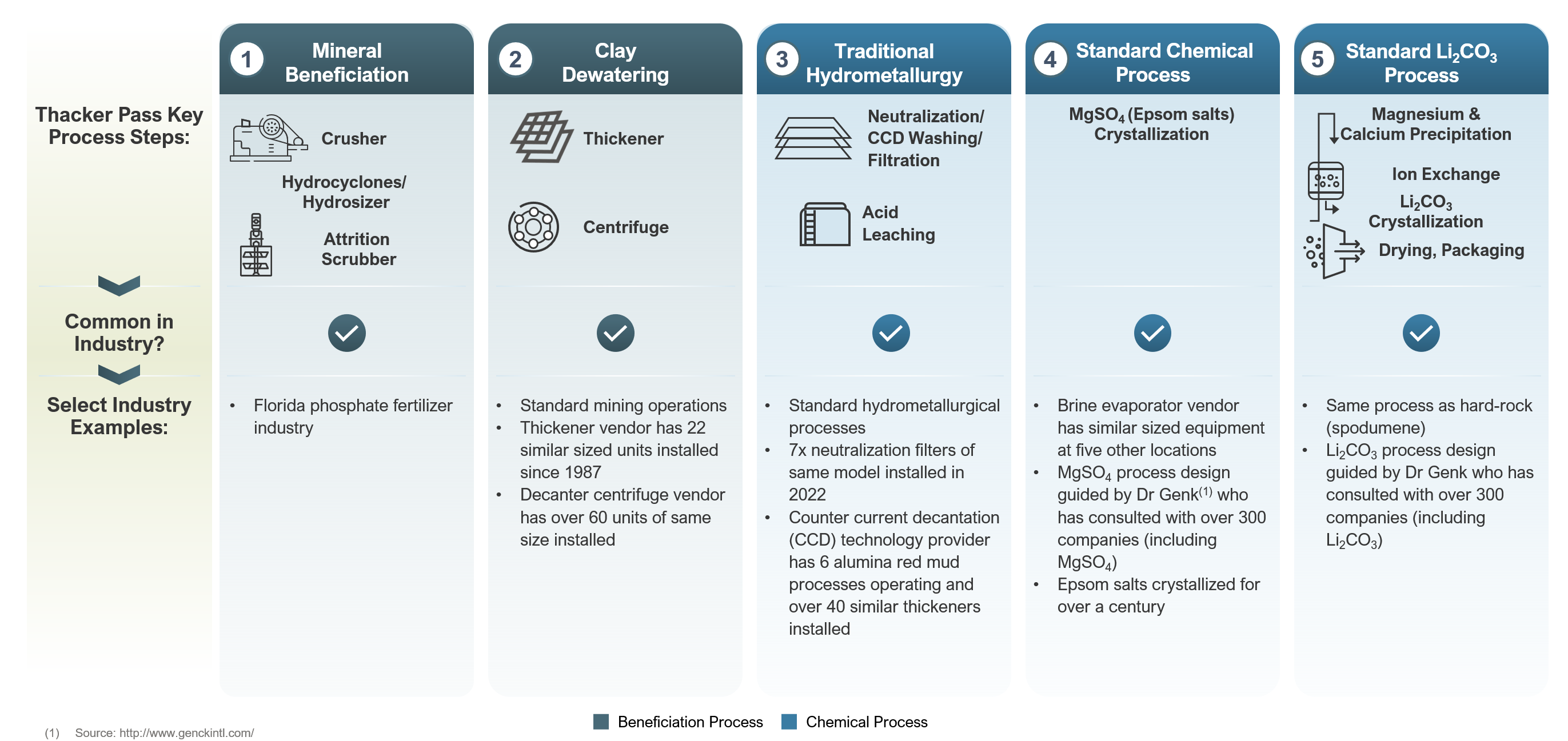
First, ore from the mine will be crushed, screened and then transferred as a slurry to the leaching circuit where sulfuric acid will be added to attack the ore and liberate the lithium from the clay. The high-grade quality of the ore allows for leaching to occur in stirred reactors (vats), specifically designed to maximize speed and efficiency of lithium dissolution, while minimizing sulfuric acid consumption. Total leaching time is estimated at three hours.
The resulting lithium-bearing solution will then go through a pH-neutralization step. Neutralization will be achieved with ground limestone during start-up and sustained with recycled alkaline solids from an upstream precipitation process during normal operation. Next, the lithium solution will undergo a crystallization step using steam and electricity from the sulfuric acid production process. Water is removed for recycling, and magnesium sulfate (Epsom salt) is produced. Any magnesium remaining in solution is removed in a second step that involves the addition of reagents to precipitate magnesium hydroxide.
Finally, soda ash will be added to the lithium bearing solution to produce a high-quality, battery-grade lithium carbonate (Li2CO3). Much of the water contained in the lithium solution will be recovered and returned to the process. The total time projected to manufacture battery- quality Li2CO3 from the ore is less than 24 hours.